Effect of therapeutic radioiodine activity on ablation response in differentiated thyroid cancer patients with cut-off serum thyroglobulin levels after 2 weeks of thyroid hormone withdrawal: a retrospective study
Article information
Abstract
This retrospective study aimed to investigate whether there was a difference in the success rate of removal of residual thyroid tissue in patients with the same cutoff serum thyroglobulin (Tg) value―measured 2 weeks after thyroid hormone withdrawal (THW)―for different radioactive iodine (RAI) activities. We identified 132 patients with papillary thyroid cancer who were treated with total thyroidectomy and RAI therapy to evaluate the efficacy of three radioactivities of I-131: 1,110, 3,700, and 5,550 MBq. Serum Tg testing was performed 1 week before RAI treatment and 2 weeks after THW (pre-Tg); the cutoff pre-Tg level was below 10 ng/mL. Stimulated Tg levels were measured on the day of I-131 administration (off-Tg). After 6 months of treatment, we compared the groups for complete ablation, defined as no uptake on a diagnostic I-131 scan, stimulated Tg level of <1.0 ng/mL, and Tg antibody level of <100 ng/mL. Ninety-five patients (72.0%) achieved complete ablation, with 57.1% (8/14), 78.2% (68/87), and 61.3% (19/31) in the 1,110 MBq, 3,700 MBq, and 5,550 MBq groups, respectively. There was no significant difference in the complete ablation rates between the three groups. In the multivariate analysis, the off-Tg level was a significant predictor of complete ablation. RAI therapy with low radioactivity (1,110 MBq) seemed sufficient for ablation in patients with papillary thyroid cancer with a pre-Tg level below 10 ng/mL. The off-Tg level is a promising and useful predictor of complete ablation after initial RAI therapy.
INTRODUCTION
Radioactive iodine (RAI) therapy has been used in differentiated thyroid cancer (DTC) as a standard treatment for the ablation of remnant thyroid tissue after total or neartotal thyroidectomy [1]. After the publication of the American Thyroid Association (ATA) guidelines in 2015, an individual approach for DTC based on a risk stratification system has emerged, resulting in a trend toward more selective use of RAI therapy [2]. The ATA guidelines recommend RAI therapy and selective use of 30-150 mCi for intermediate to high-risk patients; however, no definite consensus has been reached regarding the optimal RAI activity to achieve successful ablation. Radiation gastritis and functional impairments in the nasolacrimal and salivary glands have been reported in a significant number of patients administered RAI; these side effects are known to be related to RAI activity [3-5]. Therefore, in recent years, low-radioactivity administration has been preferred over high-radioactivity administration in RAI treatment, and studies on the treatment effect according to RAI activity have been reported [6-8].
Serum thyroglobulin (Tg) hormone measurement is an effective method for monitoring patients with DTC. Tg is produced entirely by thyroid follicular cells and is a sensitive marker of local recurrence or metastasis after removal of the residual thyroid gland [9]. Tg levels are affected by thyroid-stimulating hormone (TSH) levels >30 mU/L. The sensitivity of TSH stimulating Tg for the prediction of recurrence is 85-95% [10]. Therefore, the recurrence and metastasis of thyroid cancer are determined by measuring serum Tg following thyroid hormone withdrawal (THW) or the use of recombinant human TSH (rhTSH). Kim et al. [11] reported that Tg level immediately after thyroidectomy was a useful prognostic index for the early prediction of recurrence in patients with low-risk DTC.
Many studies have investigated treatment outcomes using serum Tg level measured 3 weeks after THW as the cutoff value. To the best of our knowledge, no study has evaluated the effectiveness of different RAI activities using serum Tg levels measured 2 weeks after THW. Therefore, this retrospective study aimed to investigate whether there was a difference in the success rate of remnant thyroid tissue removal according to RAI activity in patients with the same cutoff Tg value measured after THW for 2 weeks before treatment. Possible predictive factors for successful ablation were also evaluated.
METHODS
1. Patients
This study was approved by the Institutional Review Board (IRB No. JEJUNUH 2018-11-014-001), and the medical records of 181 patients with histopathologically confirmed classic papillary thyroid carcinoma were retrospectively reviewed. All the included patients underwent RAI treatment at our nuclear medicine department after total thyroidectomy and neck lymph node dissection between January 2015 and December 2018. Thirty-eight patients were excluded owing to loss to followup and administration of rhTSH instead of THW. Clinical characteristics were obtained from patients’ medical records, and DTC histology data were acquired from surgical pathology reports.
2. Postoperative RAI therapy protocol
All patients were prepared with a strict protocol of prolonged THW for RAI therapy, which consisted of discontinuation of levothyroxine for 3 weeks and a lowiodine diet for 2 weeks before radioiodine treatment. Tg levels after THW (pre-Tg) in all patients were measured after stopping thyroid hormone administration for 2 weeks (1 week before treatment). The administered radioactivity of RAI ablation was 30 mCi (1,110 MBq), 100 mCi (3,700 MBq), and 150 mCi (5,550 MBq), which was determined by risk classification based on the American Joint Committee on Cancer tumor-node-metastasis staging, surgical findings, histopathological findings, and patient’s condition. We administered 5,550 MBq to patients with pre-Tg levels exceeding 10 ng/mL because they were at a high risk of recurrence according to our experiments and previous reports [12]. 11 patients with a high pre-Tg level (>10 ng/mL) were excluded. In total, 132 patients were enrolled in this study.
Stimulated Tg levels at the time of RAI (off-Tg), TSH, and Tg antibody (Tg-Ab) levels were measured simultaneously in all patients on the day of I-131 administration, and postwhole-body iodine scans (WBS) were obtained 2 and 7 days after RAI therapy.
3. Assessment of RAI therapy response
Response assessment was performed 6 months after RAI therapy and was based on serum Tg measurement, neck ultrasonography (USG), and radioiodine scan. All patients underwent the same protocol as the initial RAI therapy (THW for 3 weeks and low iodine diet for 2 weeks). Stimulated Tg (after-Tg), serum TSH, and Tg-Ab levels were assessed, and WBS and neck USG were performed. Therefore, we measured stimulated Tg levels three times: 1) pre-Tg, stimulated Tg 1 week before RAI therapy; 2) off-Tg, stimulated Tg on the day of I-131 administration; and 3) after-Tg, stimulated Tg after approximately 6 months of RAI therapy. If no abnormal thyroid bed uptake was observed on the WBS, the after-Tg value was <1.0 ng/mL, USG was normal without a palpable cervical lymph node, and the Tg antibody value was 100 ng/mL. This was deemed to be successful residual thyroid removal (complete ablation, CA). We also evaluated whether the patients had recurrent disease during the long-term follow-up period, based on medical records.
4. Statistical analysis
Chi-square tests for categorical variables and Kruskal-Wallis tests for continuous variables were performed to determine whether RAI activity of 1,100, 3,700, and 5,550 MBq resulted in differences in CA and other variables between groups below the pre-Tg cutoff value. Fisher’s exact test or Mann-Whitney test was used to compare the variables between the CA and incomplete CA (ICA) groups. Logistic regression analysis of the potential predictive factors for CA was performed.
Statistical analyses were performed using MedCalc for Windows, version 12.7 (MedCalc Software, Ostend, Belgium), and P-values <0.05 were considered statistically significant.
RESULTS
1. Patient characteristics
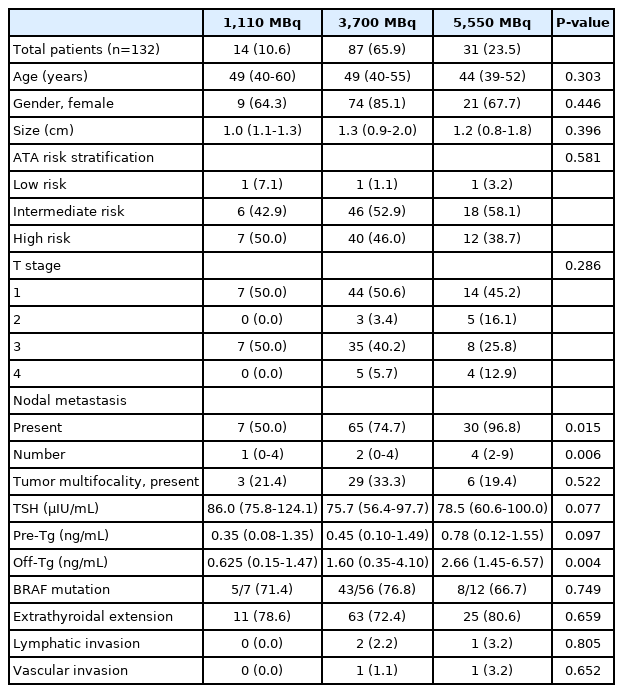
Of the 132 enrolled patients, 14 (10.6%) received 1,110 MBq of I-131, 87 (65.9%) received 3,700 MBq, and 31 (23.5%) received 5,550 MBq. The median age of the patients was 48 years and 104 (78.8%) were female. After 2 weeks of THW, the median TSH and percentile ranges were 27.4 and 13.2-37.6 μIU/mL, and the median pre-Tg level was 0.5 and 0.1-1.6 ng/mL.
The median interval between the surgery and RAI therapy was 98 days. The median values (percentile ranges) of TSH and off-Tg after 3 weeks of THW were 78.7 μIU/mL (60.3-100.3) and 1.7 ng/mL (0.4-4.3), respectively. In a follow-up evaluation 6 months after RAI therapy, the TSH and after-Tg level were 70.9 μIU/mL (34.6-100.5) and 0.1 ng/mL (0.04-0.8), respectively.
B-Raf proto-oncogene (BRAF) mutation examination was performed in 75 patients, of whom 56 (74.7%) had BRAF mutations. Lymphatic invasion was found in only three patients (2.3%), and only two (1.5%) had vascular invasion.
2. Comparison between different RAI activity groups
The patient characteristics are shown in Table 1. The presence and number of nodal metastases were significantly different among the three groups (P =0.015, P =0.006). Statistical results revealed no significant differences in age, sex, tumor size, T stage, tumor multifocality, TSH level, BRAF mutation, extrathyroidal extension, and lymphatic/vascular invasion between RAI activity groups. Although the serum off-Tg levels were significantly different among the three groups (P =0.004), the serum pre-Tg levels did not show any statistically significant differences between the groups (P =0.097).
After the initial therapy, 95 patients (72.0%) achieved CA, with 57.1% (8/14) in the 1,110 MBq group, 78.2% (68/87) in the 3,700 MBq group, and 61.3% (19/31) in the 5,550 MBq group. There was no significant difference in CA rates according to the three RAI activities (P =0.085) (Table 2).
The median follow-up period from RAI therapy was 61.9 months (range, 23.5-78.2). During the long-term follow-up, three patients in the 5,550 MBq group reported recurrence (range, 24.0-54.8 months) and were re-operated.
3. Predictors of CA
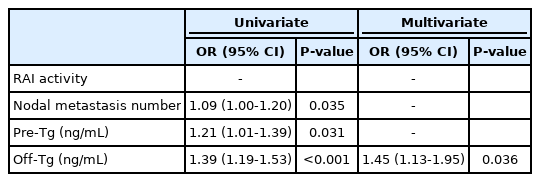
Among these variables, the number of nodal metastases (P =0.021), pre-Tg level (P =0.007), and off-Tg level (P <0.001) of the ICA group were significantly higher than that of the CA group (Table 2).
The number of nodal metastases (adjusted odds ratio [OR], 1.09; 95% confidence interval [CI], 1.00-1.20; P =0.035), serum pre-Tg level (adjusted OR, 1.21; 95% CI, 1.01-1.39; P =0.031), and off-Tg level (adjusted OR, 1.39; 95% CI, 1.19-1.53; P <0.001) were significant and independent predictors of CA in the univariate analysis. In the multivariate analysis adjusted for age and sex, the off-Tg level (adjusted OR, 1.45; 95% CI, 1.13-1.95; P =0.036) was the only significant predictor of CA (Table 3).
DISCUSSION
In this study, we demonstrated that low radioactivity iodine therapy (1,110 MBq) showed treatment efficacy similar to that of high RAI activity (3,700 MBq and 5,550 MBq) in papillary thyroid carcinoma patients with pre-Tg <10 ng/mL. The most important predictor of CA was off-Tg level at the first RAI therapy.
In recent years, there has been a trend toward a reduction in the use of RAI to decrease radiation exposure for postoperative radiation therapy. A low RAI activity of 1,110 MBq is associated with a better reduction in early side effects, such as radiation gastritis, salivary gland swelling, and discomfort, than a high RAI activity treatment. It has few late side effects such as dry mouth, eruption, and secondary cancer. Therefore, there have been many reports comparing the efficacy of low versus high RAI activity in patients with DTC; however, the optimal radioactivity remains cont roversial [13-27]. A recent meta-analysis demonstrated that the ablation rate with high RAI activity therapy was better than that with low radioactivity therapy in patients with DTC [14,15]. In contrast, most published studies have shown that low RAI activity was as effective as higher radioactivity in terms of response to therapy [16-23]. Most of these studies have been conducted by dividing patients into low, intermediate, and high-risk groups, with varying criteria for risk stratification and different opinions on the treatment effects of high radioactivity, even in studies with similar risk groups. Unlike previous studies, this is the first study to evaluate the response in patients with DTC below a certain cutoff Tg level 1 week before RAI therapy. As in most studies, there was no significant difference in RAI ablation between the low-radioactivity and high-radioactivity groups in our study. This result suggests that high RAI therapy is not helpful in patients with a pre-Tg level of <10 ng/mL. However, in previous studies, it was thought that highradioactivity therapy is necessary for the high-risk group of patients with DTC.
In the present study, the success rate was 72.0%. Previous studies have reported a wide range of CA rates on RAI therapy (10-98%) [6,22]. This might be due to different criteria for CA (Tg level and the length of time between ablation and the follow-up scan) and many other factors affecting the rate of CA, such as the extent of surgical removal and severity of the primary disease. Our relatively high CA rate could be explained by the complete surgical resection of normal thyroid tissue before RAI therapy, and a single administration of I-131 could have been sufficient for the ablation of a small amount of remnant thyroid tissue.
Only three patients reported disease recurrence over the long term, and both were in the 5,550 MBq group. None of the patients died during the follow-up period, but those who received high radioactivity tended to experience recurrence. This might be due to the disease severity, such as node metastasis or remnant thyroid tissue. Notably, there was no recurrence in the 1,110 MBq and 3,700 MBq groups, and most patients in the ICA group showed no recurrence (94.6%). From a long-term perspective, patients with a pre-Tg level of <10 ng/mL may be treated with low RAI activity, and if they have ICA after RAI therapy, they may be monitored.
Currently, several prognostic factors associated with DTC (e.g., tumor size, metastatic lymph node number, WBS uptake, and histological grade) are considered sources of valuable information regarding the likelihood of response to RAI therapy. Among these prognostic factors, the amount of remnant thyroid tissue after thyroidectomy is particularly useful in RAI therapy. In patients with DTC, the serum Tg level with TSH stimulation reflects the amount of residual malignant and normal thyroid tissue remaining and is considered a significant tumor marker [28]. Therefore, many reports have investigated the Tg level before RAI as a prognostic marker for CA [12,19,24,29,30]. Park et al. [29] demonstrated that the pre-ablative Tg level was the most useful predictor of therapeutic failure in patients with DTC. Ha et al. [30] reported that a pre-ablative Tg level with a cutoff value of 10.0 ng/mL was a promising predictor of successful remnant ablation. Consistent with previous studies, this study revealed that serum off-Tg levels, which were evaluated 3 weeks after THW, were positively associated with CA. However, pre-Tg level was not an independent prognostic factor in the multivariate analysis, although pre-Tg and off-Tg levels were significantly correlated in our analysis. This might be because the pre-Tg level obtained after 2 weeks of THW was not sufficiently stimulated to accurately reflect the remaining or residual thyroid tissues. Because we were not able to analyze the best cutoff value of off-Tg for predicting unsuccessful ablation due to the small number of patients, further research is needed.
The limitations of this study include the small number of patients, retrospective nature, and inclusion of only patients with pathologically proven papillary thyroid carcinoma. Thus, selection bias may be present. Furthermore, as this was a retrospective and non-randomized study, there was a limitation in comparing the efficacy of CA using different radioactivity levels in each group. Second, the pre-Tg cutoff value of 10 ng/mL was determined based on our individual experience. According to our empirical experiment and previous reports, patients with serum levels >10 ng/mL were considered high-risk, and 5,550 MBq was administered. However, this may also represent selection bias. Furthermore, there has been used recently rhTSH due to a convenient alternative to THW which may cause hypothyroidism symptoms. However, for stimulated Tg levels, the THW method was more accurate than rhTSH injection. Finally, this study was limited to a single hospital and only evaluated short-term responses. Therefore, multicenter and prospective studies using larger sample sizes and longer follow-up durations are needed to confirm the effectiveness of low RAI activity.
In conclusion, RAI therapy with 1,110 MBq seemed to be sufficient for ablation in patients with DTC with a pre-TG level <10 ng/mL 2 weeks after THW. Low radioactivity avoids unnecessary radiation exposure, reduces the isolation period, has a lower cost, and reduces adverse effects. The off-Tg level after THW is a promising and useful predictive factor for CA after initial RAI therapy.
Acknowledgements
This work was supported by a research grant from Jeju National University Hospital in 2018.